
Used in jet fuel, and HCO 3 −, used in antacids. Used with calcium ions as a dough conditioner, ClO 4 −, Write names that correspond to the following formulas for The two H + ions neutralize two of the three minus The dihydrogen part of the name indicates that two H + ions have Hypophosphite has two less oxygen atoms than phosphate, PO 4 3 −. Bromate is BrO 3 −.īromite has one less oxygen atom than bromate. (2) dihydrogen hypophosphite ion, used with manganese(II) ions as a foodġ. (1) bromite ion, used in the production of cloth Write formulas that correspond to the following names for This could be called by its nonsystematic name, bisulfate, but it is preferable to use the systematic name, which shows that one H + ion has been added to sulfate, SO 4 2. Alternatively, you can think of it that the sum of the. PO 4 3 − is phosphate, and this ion has one lessĢ. The oxidation state of a simple ion like hydride is equal to the charge on the ion - in this case, -1. Write names that correspond to the following formulas for polyatomic Systematic and Nonsystematic Names for Some Polyatomic Ions You shouldĪvoid using these less accepted names, but because many people still use You know that IO 4 - is periodate, because it has oneĪlso have nonsystematic names that are often used (Table 3). You know that NO 2 - is nitrite, because it has one The anion with two less oxygen atoms than the (root)ateĭifferent number of oxygen atoms Relationship The anion with one fewer oxygen atom than the (root)ateĪnion is named with -ite on the end of the root. 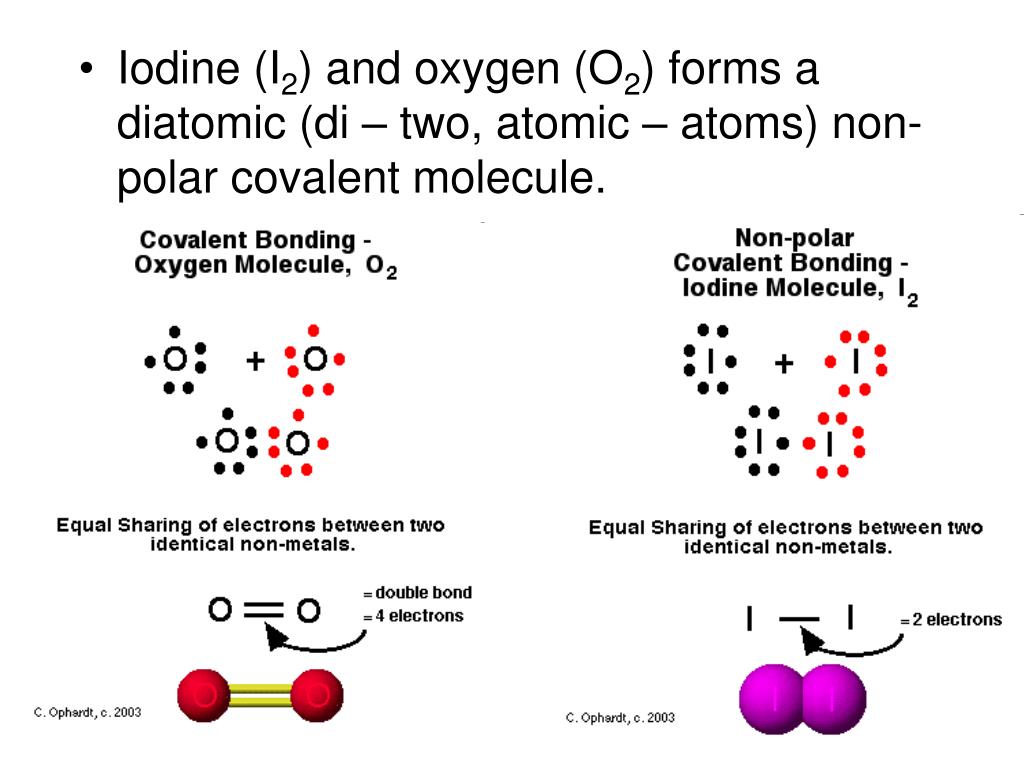
The anion with one more oxygen atom than the (root)ate The names of the other possible oxyanions are determined as Oxyanions has a name with the form (root)ate. The most common of the chlorine oxyanionsįact, you will generally find that the most common of an element’s Of oxyanions, the charge remains the same only the number of oxygen atoms For example, chlorine can combine with oxygen in four ways to formįour different oxyanions: ClO 4 −, ClO 3 −,ĬlO 2 −, and ClO −. 
Ions that contain oxygen), each containing a different number of oxygenĪtoms. Some elements are able to form more than one oxyanion (polyatomic
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
